|
7/27/2023 0 Comments Carbon disulfide cs2Here, the bond angles form an angle of 180 degrees. There are two sigma bonds formed, thus steric number=2, sp hybridization.Īs discussed earlier, CS2 has a linear shape. The octet has been fulfilled by the help of sharing valence electrons among the two sulfur and the carbon atoms, therefore here there is no lone pair. Steric Number= No of sigma bonds + No of pi lone pairs( of the central atom, here Carbon). It signifies that the hybridization type is sp.Īlso, we can calculate the steric number. Here is a formula that can calculate the hybridization value H=0.5(V+M-C+A) The electrons of 2s and 2p orbitals of C form the double bonds and thus they result in sp hybridization. Just in the above section, while discussing the Lewis Structure we learned that in CS2 two double bonds are formed and eight of the 16 valence electrons are used up for this purpose. S ground state configuration: 1s2 2s2 2p6 3s2 3p4 If a central atom( here C) has two valence electron density regions surrounding it, then it exhibits sp hybridization.Ĭ ground state configuration: 1s2 2s2 2p2 Let us first make you go through the theoretical standpoint to make things easier!Īn ‘s’ orbital will overlap with a ‘p’ orbital to create sp hybridization. This process of hybridization is one of the vital concepts of bonding and can be explained in two different methods- one with the help of theory and the other one depending on formulae. Now, what is hybridization? Do you know that atoms use hybrid orbitals and not atomic orbitals to form chemical bonds in a molecule? This is how we draw a complete Lewis Structure of Carbon disulfide. Now, we need to check the formal charge following the given mentioned formula. We are therefore changing the lone pairs into double bonds as per necessity.Ħ. We can easily mend this issue by sharing the electrons around sulfur and bringing them alongside carbon, thus 2 valence electrons from each sulfur atom towards carbon will solve the problem here.ĥ. But, carbon is the only one left with only four electrons surrounding it. We can check and find out that both the sulfur atoms have fulfilled their octet rule here. The skeleton diagram of CS2 is drawn below.Ĥ. According to Step 2, carbon is the least electronegative having the highest bonding sites. Total valence electrons is CS2 = 12+4 = 16.Ģ. Sulfur(S) belonging to Group 6 has 6 valence electrons.ĬS2 has two S atoms, hence, the valence electrons in sulfur here are 6*2=12. Therefore, the number of valence electrons in the Carbon atom =4. Carbon belongs to Group 4 of the periodic table. Let us know see how we can draw the Lewis Structure for CS2ġ. Here is how we can easily calculate the formal charge: (A formal charge, also abbreviated as FC is the charge that is assigned to an atom of a molecule when we assume that chemical bonds are always shared equally between atoms inside a molecule.) Step 6: The final step of Lewis diagram formation is to verify whether all the atoms are in their lowest possible formal charge. If there is a requirement of multiple bond formation, we have to create double or triple bonds as per requirements to fulfill the octet rule. Step 5: In this step, we have to check whether there are bonds left to be formed. The advisable decision is to finish the electronegative ones first before starting with the electropositive atoms. Step 4: Next, our task is to complete the octet of the atoms around each of the outer ones with the remaining electrons. Step 3: Now, we need to draw a skeleton diagram having the presence of single bonds. 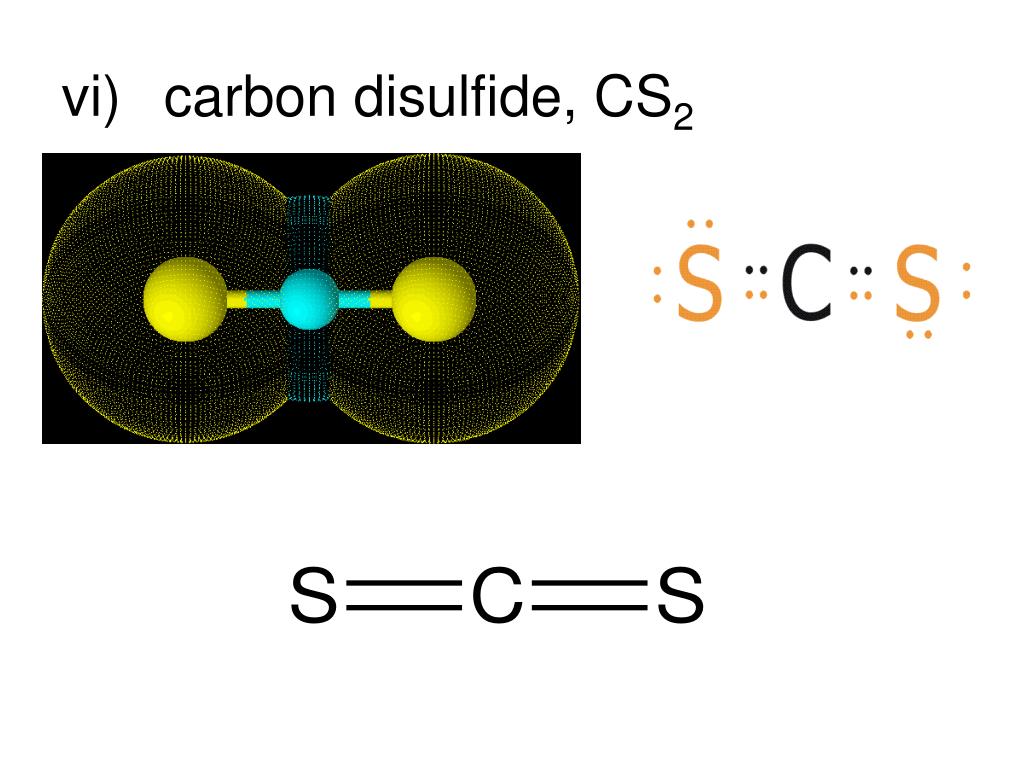
As per the trend, it is likely to decrease down a group. We can determine the electronegativity value by browsing through the periodic table. bonding sites ( in other words, least electronegative) is the central atom. 
Usually, the one with the highest valence i.e. Step 2: The second step is based on finding the central atom of the molecule. While the positive ‘+’ sign indicates the loss of electrons i.e loss of negative charge, the negative ‘-’ sign is to denote the gain of electrons. (A valence electron is a name given to the outer shell electron of an atom that takes part in the creation of a chemical bond).Ī very important point to be noted here is the role of the signs ‘+’ and ‘-’. Step 1: The very first step towards drawing the structure of a molecule is to decipher the total number of valence electrons.  
Thus, to have a comprehensive idea about CS2 Lewis Structure, let us go through each step clearly and systematically. It depends on the octet rule concept and is an extension of the electron dot diagram. Lewis Structure is one of the key terminologies to understand the chemical bonding of a molecule since it represents the molecular structure. If not, here’s a brief explanation of the above-mentioned topic. If you are a student of chemistry, it is almost obvious that you are aware of the term ‘Lewis Structure”.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
